Read This First
SarcoSupport Case Study: Observed Sarcoid Regression Following Nutritional Support
This case study documents observations from a single horse over a defined period while a complementary feed was included in the daily ration. Individual cases do not predict outcomes in other horses. It is shared to explain how SarcoSupport was developed and to provide transparency around what was observed, when, and under what conditions.
This is not a clinical trial, veterinary diagnosis, or a claim of treatment, prevention, or cure. Sarcoids vary widely between horses, and outcomes differ. The information below is presented to inform understanding, not to predict results. The observations recorded here describe what happened in one horse and do not establish cause and effect. Sarcoids can occasionally regress without intervention, and many factors may influence their progression.
SarcoSuppor is a complementary feed delivering naturally occurring phytonutrients and antioxidants, formulated over many years to support normal immune and cellular processes through nutrition. This case study should be read as contextual information to support informed decision-making alongside appropriate professional advice where needed.
Case Overview
This case study documents the observed progression and eventual detachment of a suspected fibroblastic sarcoid in a horse over a 12-week period, during which a complementary feed delivering naturally occurring phytonutrients and antioxidants (SarcoSupport) was included in the horse’s daily ration. No veterinary, topical, caustic, or surgical interventions were used during the observation period. This account is presented as a factual record of a single case and does not establish causation or predict outcomes in other horses.
Timeline
Case summary and observational timeline
Key case details and week-by-week observations for the 12-week period during which SarcoSupport was included in the daily ration.
Case ID: 001
Observation period: 27 July 2025 – 17 October 2025 (82 days)
Animal: Horse (gelding)
Approximate weight: 500–550 kg
General health: Good; no known systemic illness
Diet: Forage-based diet with balancer and chaff
Concurrent treatments: None
Sarcoid description at initial observation
Suspected type: Fibroblastic sarcoid
Location: Axillary region, adjacent to the front leg (armpit area)
Characteristics:
• Raised, pedunculated lesion
• Approx. 2.5–3 cm diameter
• Partially raw surface with intermittent crusting
• Located in a high-movement area subject to friction
The lesion was not biopsied due to the recognised risk of local spread associated with biopsy of sarcoids.
Week-by-week observational record
Weeks 1–3
The lesion remained stable in size. The surface appeared intermittently moist with areas of dry crusting. No marked increase in inflammation or discomfort was observed.
Weeks 4–7
Progressive surface drying was noted. The lesion began to show increased crust formation and subtle reduction in apparent volume.
Weeks 8–11
Clear narrowing at the base of the lesion became visible. The sarcoid appeared less vascular, with no signs of secondary infection or acute inflammation.
Week 12
The lesion detached spontaneously. The underlying tissue appeared clean, with no excessive bleeding or abnormal granulation. Surrounding skin tone was normal. Photographic records were taken at regular intervals throughout the observation period.
Post-detachment monitoring
Following detachment, the site was monitored without intervention. No immediate regrowth or adverse tissue response was observed in the 5 months following separation.
Feeding protocol
Nutritional intervention used in this case
The information below describes how SarcoSupport was incorporated into the daily ration for this individual horse. It does not constitute a recommendation or prescription and should be considered alongside individual requirements and professional guidance where appropriate.
Product: SarcoSupport Standard
Classification: Complementary feed for horses
Form: Freeze-dried plant-based powder
Daily inclusion rate: Approximately 25 g per day
Method of feeding: Mixed into daily feed ration
No topical application was used after a brief early attempt was discontinued. No other supplements, medications, or veterinary treatments were introduced during the observation period.
SarcoSupport is a complementary feed and is not a veterinary medicinal product. It does not contain medicinal substances and is not intended or presented to treat, prevent, or cure disease.
Observations
Factual record of lesion behaviour over time
Across the 12-week observation period, the lesion showed a transition from a moist, partially raw surface to a progressively drier, more crusted appearance, followed by visible narrowing at its base and eventual spontaneous detachment.
At no point during the observation period were signs of acute inflammation, secondary infection, or marked discomfort recorded. The surrounding skin remained within normal limits on visual assessment.
The post-detachment phase was characterised by a clean underlying tissue surface, absence of excessive bleeding, and no abnormal granulation observed. Over a further 5-month monitoring period, no immediate regrowth or adverse local tissue response was noted.
Regulatory context
SarcoSupport is a complementary feed for horses. It does not contain medicinal substances and is not presented as a veterinary medicinal product.
Sarcoids are complex conditions with variable outcomes. This case study records an individual observation and does not claim to diagnose, treat, prevent, or cure disease, nor does it establish causation or predict outcomes in other animals.
Horse owners should consider nutritional strategies as part of overall management decisions and consult appropriately where veterinary assessment is required.
Interpretation
How this single case is understood
Sarcoids are known to have variable and unpredictable natural histories, including occasional spontaneous regression. This case documents an instance in which regression and detachment were observed during a period of dietary inclusion of a complementary feed providing concentrated plant phytonutrients and antioxidants.
This observation does not establish a causal relationship. It may, however, be relevant to the nutritional context within which regression was observed. Further structured observation across a broader population would be required before any general conclusions could be drawn.
This case is therefore best understood as one detailed, real-world observation contributing to a wider body of practical experience, rather than as definitive evidence of effect.
Why this case is shared
This case is shared to provide transparency around the origin of SarcoSupport and the process by which its formulation has been shaped over time. The formulation has been informed by approximately ten years of iterative development, practical experience, and nutritional refinement.
The case documents observed outcomes in a real-world setting and reflects a commitment to evidence-led formulation, careful observation, and responsible communication. It is intended to support informed decision-making alongside appropriate veterinary and professional input.
Photo gallery
Images from the observation period
Photographs were taken at regular intervals to document the appearance of the lesion over time. The images below are some of the pictures taken during the observation timeline. Images are provided for visual context only and should not be interpreted as establishing cause and effect.
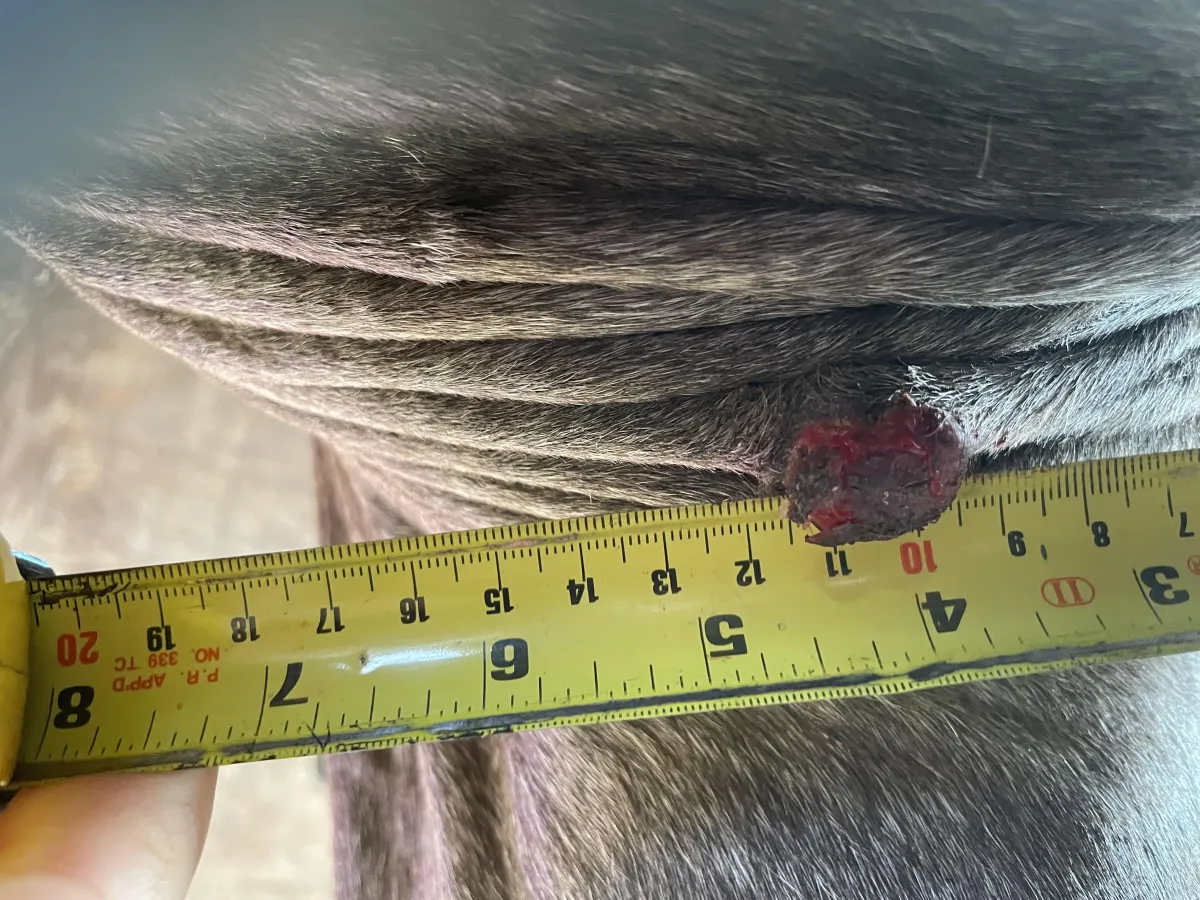
Week 0 – Initial presentation. Raised, pedunculated lesion with partially raw surface and intermittent crusting in the axillary region.
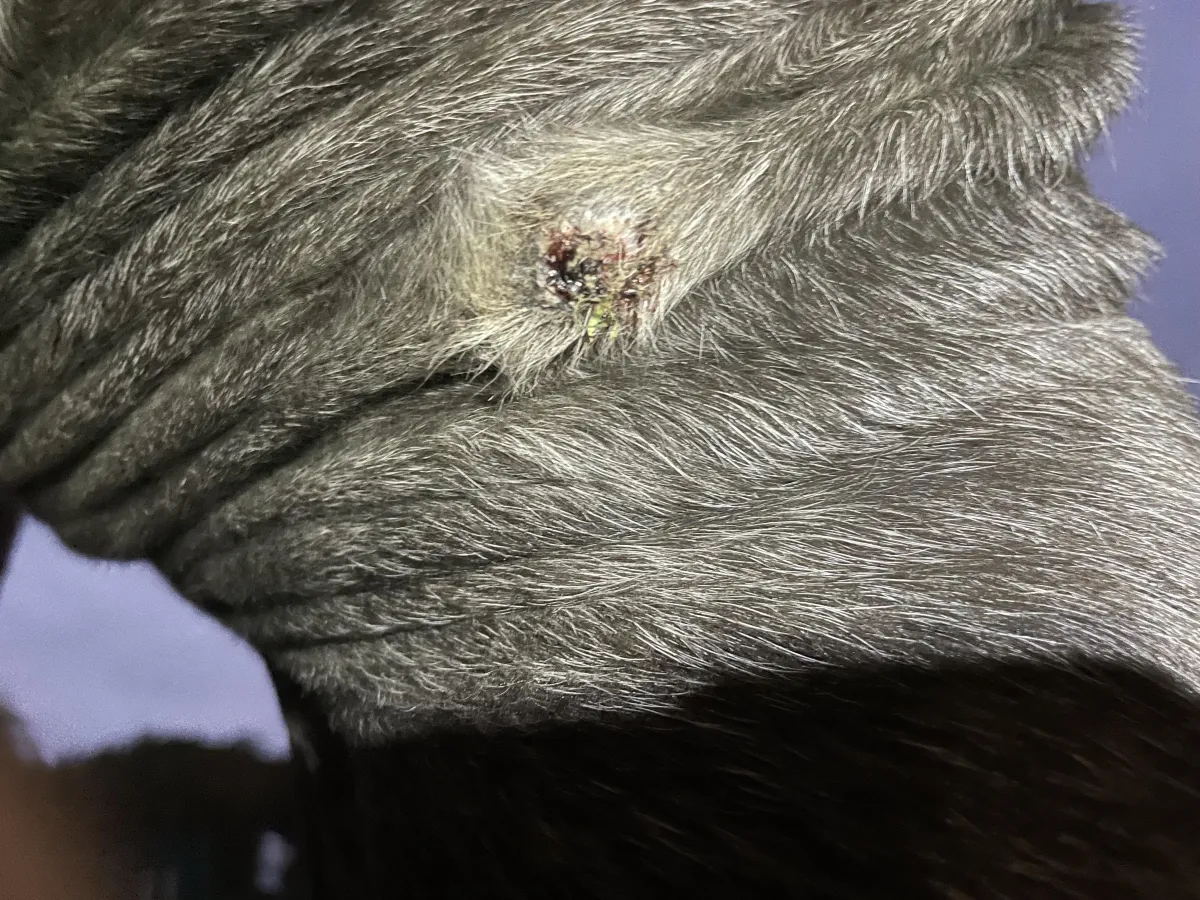
Week 12 and early post-detachment period – Clean underlying tissue with no excessive bleeding or abnormal granulation. Surrounding skin tone within expected limits.
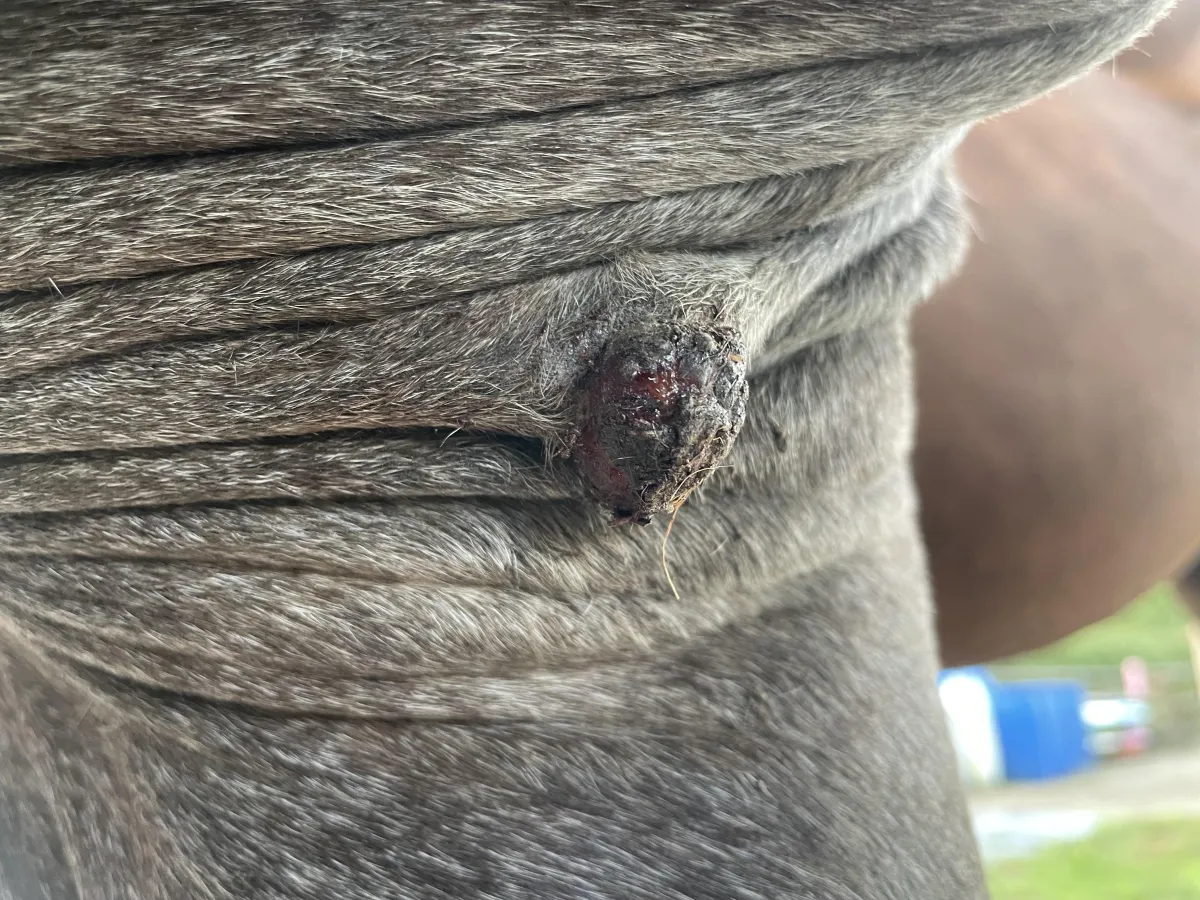
Weeks 6–9 – Increased crust formation and visible narrowing at the base of the lesion. No secondary infection observed.
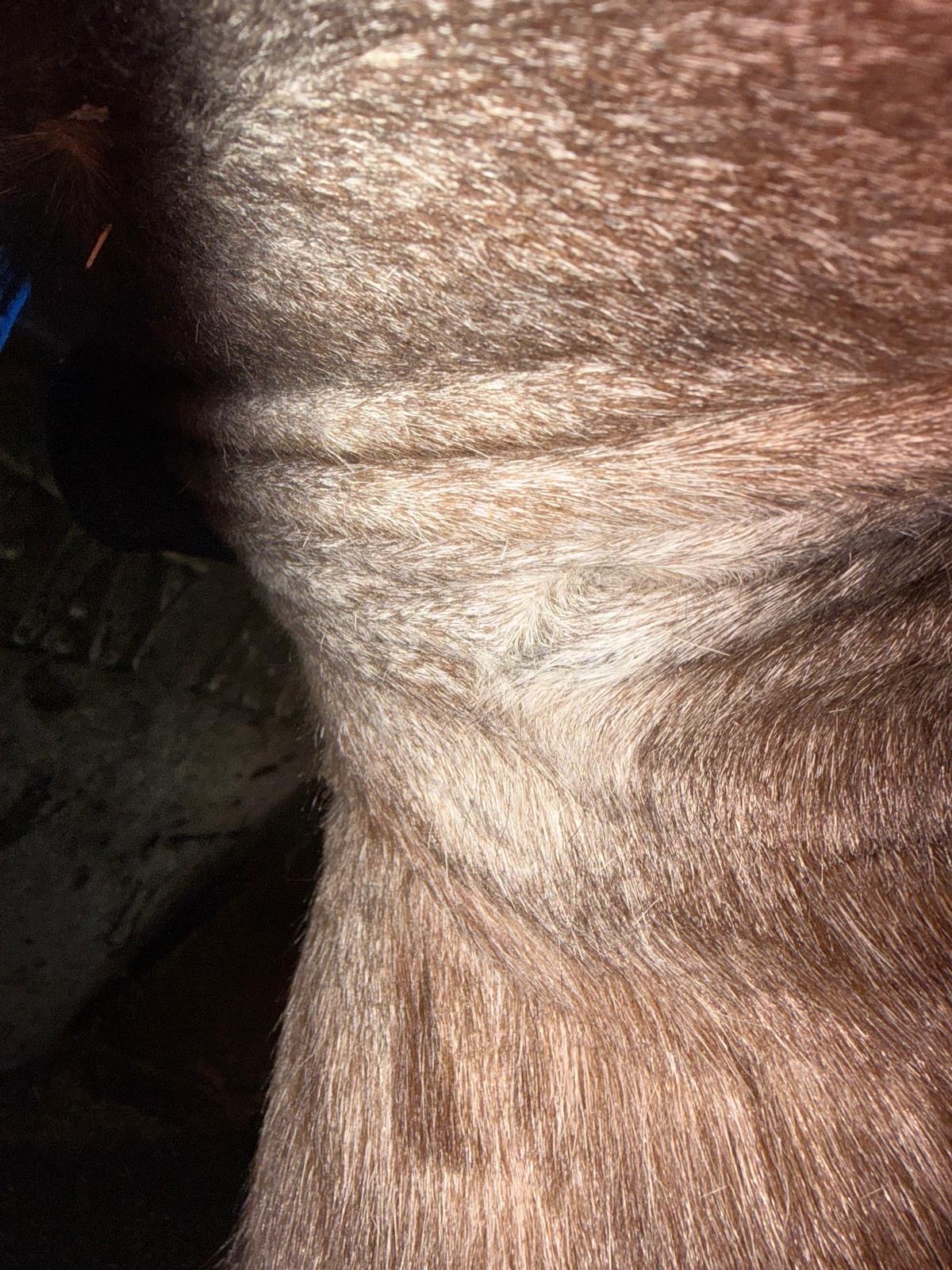
Month 5 after detachment, skin and site of the sarcoid appear normal with no obvious granulation.
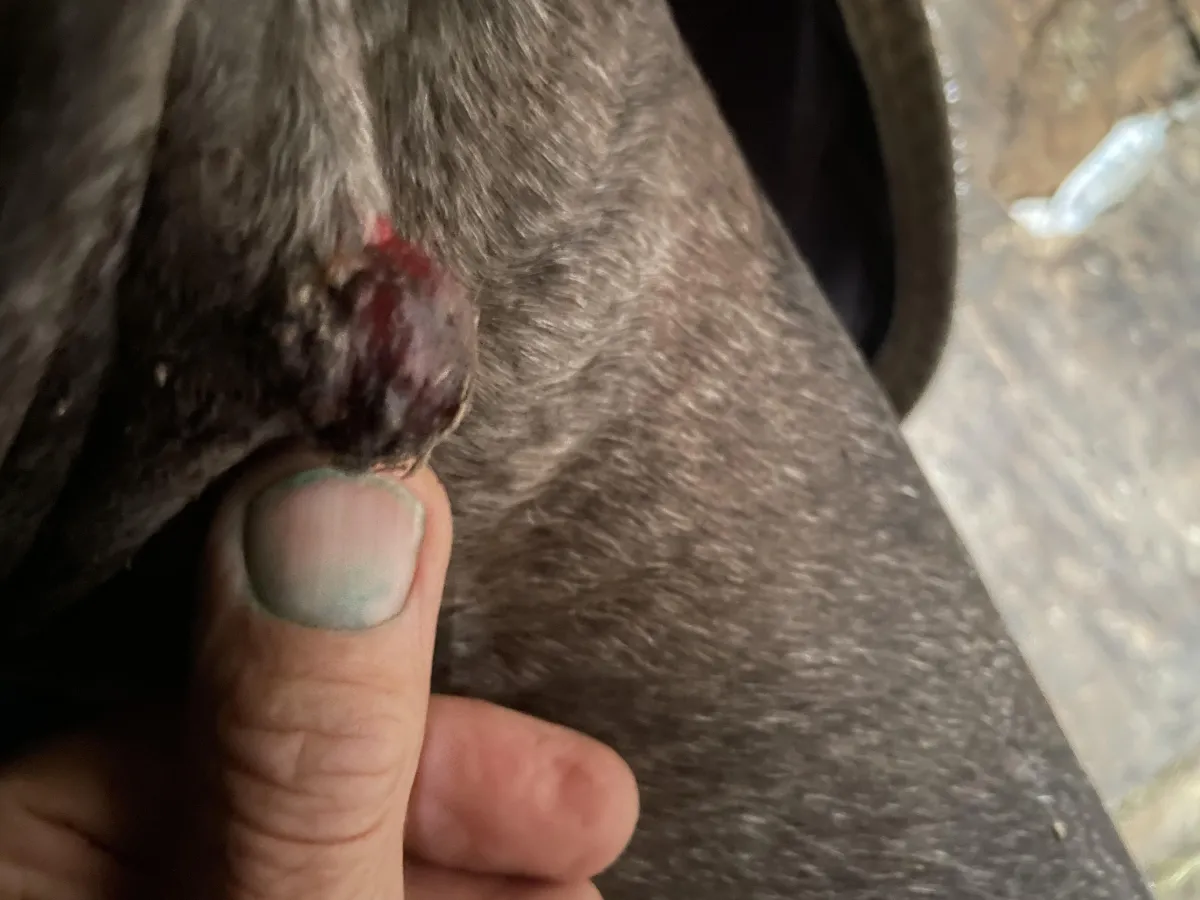
Week 10 base of the lesion continues to narrow. Still no secondary infection observed.
